Selvol™ Polyvinyl Alcohol Product Information
Manufacturers commonly use Selvol polyvinyl alcohol (PVOH) for adhesives. However, the industry and agriculture sectors also use it in emulsion polymerization, films and packaging, as well as in oil field cementing, paper, textiles, ceramics and seed coatings.
As a top PVOH supplier, SEKISUI continually develops new applications for and products featuring PVOH.
Manufacturers make Selvol PVOH from vinyl acetate monomer (VAM) via a multistep process. They polymerize VAM into polyvinyl acetate and then convert it to PVOH.
Would you like more information? Learn about Selvol PVOH here.
Grades
Selvol PVOH grades offer incredible versatility in one core product. In fact, it’s the most versatile PVOH product we offer. The key to working ideally with Selvol PVOH is finding the grade that suits your needs.
Standard Grades
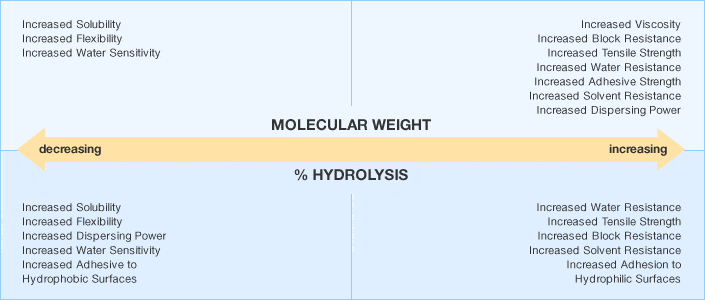
Each Selvol PVOH grade varies in molecular weight and degree of hydrolysis.
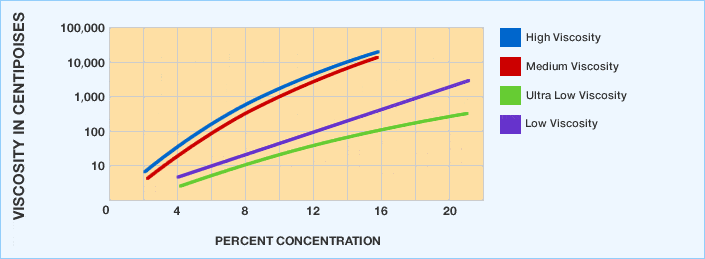
We generally express molecular weight in terms of solution viscosity. First, we classify the viscosities as ultra low, low, medium and high. We commonly denote the degree of hydrolysis as super, fully, intermediately and partially hydrolyzed. Additionally, we offer a wide range of standard grades.
In addition to SEKISUI Specialty Chemicals’ standard product line, we provide multiple specialty grades. These include polymerization, fine particle size and tackified PVOHs:
- Selvol PVOHs 805, 823 and 840 are improved versions of our standard polymerization grades (Selvol PVOHs 205, 523 and 540, respectively). These products offer many advantages in emulsion polymerization applications, including better water solubility and lower foaming.
- Selvol PVOH S-grades are similar to our standard grades. However, they have a fine particle size so that more than 99% of the product passes through a U.S.S. 80-mesh screen. These grades are available commercially as Selvol PVOHs 203S, 205S, 523S and 540S.
- Tackified Selvol PVOHs are products of the controlled boration of super-hydrolyzed and fully hydrolyzed grades. These borated alcohols yield viscous, aqueous solutions. These solutions also have a tailored degree of tack and significantly reduce penetration when applied to surfaces such as paper.
Selvol Polyvinyl Alcohol Solutions
At SEKISUI Specialty Chemicals, we provide premade solutions of some of our standard grades. A Selvol PVOH solution eliminates the dissolution step and creates distinct advantages:
- Ready-to-use, improved solution consistency
- Lower energy costs
- Lower processing costs (no cooking required)
Please note Selvol PVOH solutions are available in the U.S. only.
Product Availability
Selvol PVOH standard and specialty grades are available globally. SEKISUI Specialty Chemicals supports sales with an in-region, worldwide workforce.
Contact us for more information.
Selvol E PVOH
The Selvol E PVOH line has a wide variety of grades to meet your needs. Overall, Selvol E PVOH comprises standard grades, which we denote by their degree of hydrolysis: fully, intermediately or partially. We manufacture the Selvol E PVOH product line in Tarragona, Spain, and the line primarily supports the European market.
Please see the product literature page to download technical data sheets.
Explore the chart below to learn the line’s typical properties and applications.